- feeds
- popular
- recent
- reader
- about
- story
- technologies
- what is rss
- connect
- dmca
- contact
- Feed Preview Med-Chemist
FDA Approves Tepylute (thiotepa) Ready-to-Dilute Injectable Formulation to Treat Breast Cancer and Ovarian Cancer

Shorla Oncology (‘Shorla’), a U.S.-Ireland specialty pharmaceutical company, announced today that the U.S. Food and Drug Administration (FDA) has approved the company’s New Drug Application...
https://www.med-chemist.com/2024/10/fda-approves-tepylute-thiotepa-ready-to.html
FDA Approves Ohtuvayre (ensifentrine) for the Maintenance Treatment of Chronic Obstructive Pulmonary Disease (COPD)

Verona Pharma plc (Nasdaq: VRNA) (“Verona Pharma” or the “Company”), announces the US Food and Drug Administration (“FDA”) approved Ohtuvayre (ensifentrine) for the maintenance trea...
https://www.med-chemist.com/2024/10/fda-approves-ohtuvayre-ensifentrine-for.html
FDA Approves Sofdra (sofpironium) Topical Gel for the Treatment of Primary Axillary Hyperhidrosis

Clinical dermatology company, Botanix Pharmaceuticals Ltd. (ASX:BOT, Botanix or the Company), is pleased to announce the US Food and Drug Administration (FDA) approval of Sofdra™ (sofpironium)...
https://www.med-chemist.com/2024/10/fda-approves-sofdra-sofpironium-topical.html
FDA Approves Vigafyde (vigabatrin) as the First and Only Ready-to-Use Vigabatrin Oral Solution

Pyros Pharmaceuticals, Inc. (Pyros or the Company), a leader in the development of enhanced specialty pharmaceuticals for rare diseases, announced today that the U.S. Food and Drug Administration...
https://www.med-chemist.com/2024/10/fda-approves-vigafyde-vigabatrin-as.html
FDA Grants Accelerated Approval to Iqirvo (elafibranor) for the Treatment of Primary Biliary Cholangitis

Ipsen (Euronext: IPN; ADR: IPSEY) today announced that the U.S. Food and Drug Administration (FDA) has granted accelerated approval for Iqirvo (elafibranor) 80 mg tablets for the treatment of p...
https://www.med-chemist.com/2024/10/fda-grants-accelerated-approval-to.html
FDA Approves Lumisight (pegulicianine) Optical Imaging Agent to Illuminate Residual Breast Cancer Post-Lumpectomy
Lumicell, Inc., a privately held company focused on developing innovative fluorescence-guided imaging technologies for cancerous tissue detection during surgery, today announced the U.S. Food & D...
https://www.med-chemist.com/2024/10/fda-approves-lumisight-pegulicianine.html
FDA Approves Xromi (hydroxyurea) Oral Solution for Use in Pediatric Patients with Sickle Cell Anemia

The U.S. Food and Drug Administration (FDA) has approved Xromi, an oral solution formulation of hydroxyurea indicated to reduce the frequency of painful crises and reduce the need for blood tr...
https://www.med-chemist.com/2024/10/fda-approves-xromi-hydroxyurea-oral.html
FDA Approves Rytelo (imetelstat) for the Treatment of Adult Patients with Lower-Risk MDS with Transfusion-Dependent Anemia

Geron Corporation (Nasdaq: GERN), a commercial-stage biopharmaceutical company aiming to change lives by changing the course of blood cancer, today announced that the U.S. Food and Drug Admin...
https://www.med-chemist.com/2024/09/fda-approves-rytelo-imetelstat-for.html
FDA Grants Accelerated Approval for Ojemda (tovorafenib) for Relapsed or Refractory BRAF-altered Pediatric Low-Grade Glioma

Day One Biopharmaceuticals, Inc. (Nasdaq: DAWN) (“Day One” or the “Company”), a commercial-stage biopharmaceutical company dedicated to developing and commercializing targeted therapies f...
https://www.med-chemist.com/2024/09/fda-grants-accelerated-approval-for_0541614428.html
FDA Approves Onyda XR (clonidine hydrochloride) Non-Stimulant Liquid Treatment for ADHD

Tris Pharma, Inc. (Tris), a commercial-stage biopharmaceutical company focused on attention deficit hyperactivity disorder (ADHD), pain, addiction and neurological disorders, today announced the ...
https://www.med-chemist.com/2024/09/fda-approves-onyda-xr-clonidine.html
FDA Approves Risvan (risperidone) for the Treatment of Schizophrenia

Today, Laboratorios Farmacéuticos Rovi, S.A. (“ROVI” or the “Company”) has announced that the U.S. Food and Drug Administration (FDA) has authorized the marketing of Risvan® (Risperido...
https://www.med-chemist.com/2024/09/fda-approves-risvan-risperidone-for.html
FDA Approves Zevtera (ceftobiprole medocaril sodium) for Bacteremia, Skin and Skin Structure Infections, and Pneumonia

Basilea Pharmaceutica Ltd, Allschwil (SIX: BSLN), a commercial-stage biopharmaceutical company committed to meeting the needs of patients with severe bacterial and fungal infections, announced to...
https://www.med-chemist.com/2024/09/fda-approves-zevtera-ceftobiprole.html
FDA Approves Myhibbin (mycophenolate mofetil) Oral Suspension for Prophylaxis of Organ Rejection

Azurity Pharmaceuticals, a pharmaceutical company focused on developing innovative dose forms and formulations of products to serve the needs of overlooked patients, announced today that the...
https://www.med-chemist.com/2024/09/fda-approves-myhibbin-mycophenolate.html
FDA Approves Libervant (diazepam) Buccal Film for the Treatment of Seizure Clusters in Pediatric Patients Ages 2-5 Years

Aquestive Therapeutics, Inc. (NASDAQ: AQST) (“Aquestive” or the “Company”), a pharmaceutical company advancing medicines to bring meaningful improvement to patients' lives through innovat...
https://www.med-chemist.com/2024/09/fda-approves-libervant-diazepam-buccal.html
FDA Approves Pivya (pivmecillinam) for the Treatment of Uncomplicated Urinary Tract Infections
Today, the U.S. Food and Drug Administration approved Pivya (pivmecillinam) tablets for the treatment of female adults with uncomplicated urinary tract infections (UTIs) caused by susceptible i...
https://www.med-chemist.com/2024/09/fda-approves-pivya-pivmecillinam-for.html
FDA Approves Voydeya (danicopan) as Add-On Therapy for the ...Extravascular Hemolysis in Paroxysmal Nocturnal Hemoglobinuria

Voydeya (danicopan) has been approved in the US as an add-on therapy to eculizumab or eculizumab for the treatment of extravascular hemolysis (EVH) in adults with paroxysmal nocturnal hemoglobin...
https://www.med-chemist.com/2024/09/fda-approves-voydeya-danicopan-as-add.html
FDA Approves Vafseo (vadadustat) for the Treatment of Anemia due to Chronic Kidney Disease in Adult Patients on Dialysis

Akebia Therapeutics, Inc. (Nasdaq: AKBA), a biopharmaceutical company with the purpose to better the lives of people impacted by kidney disease, today announced that the U.S. Food and Drug Admini...
https://www.med-chemist.com/2024/09/fda-approves-vafseo-vadadustat-for.html
FDA Approves Duvyzat (givinostat) for Duchenne Muscular Dystrophy

Italfarmaco S.p.A. announced today that the U.S. Food and Drug Administration (FDA) has approved Duvyzat™ (givinostat), a novel histone deacetylase (HDAC) inhibitor, for the treatment of pa...
https://www.med-chemist.com/2024/09/fda-approves-duvyzat-givinostat-for.html
FDA Approves Opsynvi (macitentan and tadalafil) for Adults with Pulmonary Arterial Hypertension
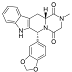
Johnson & Johnson today announced that the U.S. Food and Drug Administration (FDA) has approved Opsynvi® – a single-tablet combination of macitentan, an endothelin receptor antagonist (ERA),...
https://www.med-chemist.com/2024/09/fda-approves-opsynvi-macitentan-and.html
FDA Approves Rezenopy (naloxone hydrochloride) Nasal Spray for the Emergency Treatment of Opioid Overdose

The U.S. Food and Drug Administration has approved Rezenopy (naloxone hydrochloride) nasal spray 10 mg for emergency treatment of known or suspected opioid overdose, as manifested by respirator...
https://www.med-chemist.com/2024/09/fda-approves-rezenopy-naloxone.html
FDA Approves Tryvio (aprocitentan) for the Combination Treatment of Resistant Hypertension

Idorsia Pharmaceuticals U.S. Inc. announced the US Food and Drug Administration (FDA) approval of Tryvio™ (aprocitentan) for the treatment of hypertension in combination with other antihype...
https://www.med-chemist.com/2024/09/fda-approves-tryvio-aprocitentan-for.html
FDA Grants Accelerated Approval for Rezdiffra (resmetirom) ...Patients with Noncirrhotic Nonalcoholic Steatohepatitis (NASH)

Madrigal Pharmaceuticals, Inc. (NASDAQ:MDGL), a biopharmaceutical company focused on delivering novel therapeutics for nonalcoholic steatohepatitis (NASH), announced the U.S. Food and Drug Admi...
https://www.med-chemist.com/2024/09/fda-grants-accelerated-approval-for.html
FDA Approves Xolremdi (mavorixafor) for Use in Patients with WHIM Syndrome

X4 Pharmaceuticals (Nasdaq: XFOR), a company driven to improve the lives of people with rare diseases of the immune system, today announced that the U.S. Food and Drug Administration (FDA) has ap...
https://www.med-chemist.com/2024/09/fda-approves-xolremdi-mavorixafor-for.html
FDA Approves Exblifep (cefepime/enmetazobactam) for the Treatment of Complicated Urinary Tract Infections,

Allecra Therapeutics (“Allecra”), a biopharmaceutical company developing novel therapies to combat antibiotic resistance, announced today that the U.S. Food and Drug Administration (F...
https://www.med-chemist.com/2024/09/fda-approves-exblifep.html
FDA Approves Fruzaqla (fruquintinib) for Previously Treated Metastatic Colorectal Cancer
Takeda (TSE:4502/NYSE:TAK) today announced that the U.S. Food and Drug Administration (FDA) has approved Fruzaqla ™ (fruquintinib), an oral targeted therapy for adults with metastatic colorect...
https://www.med-chemist.com/2024/09/fda-approves-fruzaqla-fruquintinib-for_01548910670.html
